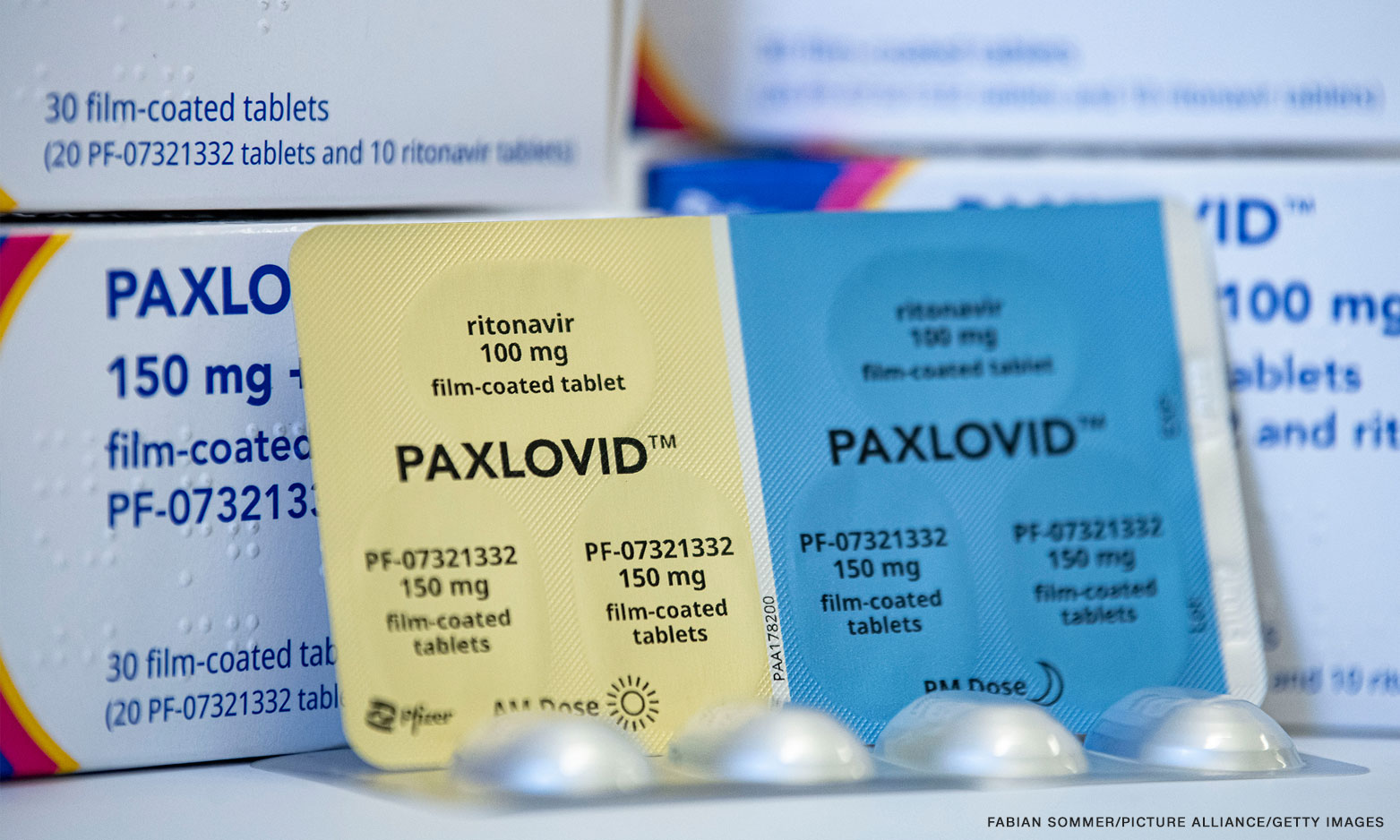
Metro Manila (CNN Philippines, March 11) — The Food and Drug Administration has approved the emergency use authorization for Pfizer’s oral antiviral COVID-19 drug Paxlovid, the agency’s officer-in-charge Oscar Gutierrez said on Friday.
This is the second oral COVID-19 treatment approved by the drug regulatory agency, following the EUAs granted to different brands of molnupiravir.
“EUA po iyon, so dalawa na ang oral antiviral treatment natin against COVID-19: Paxlovid and molnupiravir,” he said in the government-hosted Laging Handa briefing.
[Translation: That is emergency use authorization. We now have two oral antiviral treatments against COVID-19: Paxlovid and molnupiravir.
On the FDA website, Paxlovid is advised for adult patients with mild to moderate symptoms who are at high risk for progression to severe COVID-19.
The Department of Health has been in talks with Pfizer to procure Paxlovid tablets.
Paxlovid, which combines two antiviral drugs nirmatrelvir and ritonavir, is the first antiviral drug approved by the US FDA to treat COVID-19 in high-risk individuals. According to Pfizer’s latest study, this drug is able to cut the risk of hospitalization or death by 89% if given to high-risk adults within a few days of their first symptoms.